Inside ASFaVIP projet:
Advancing the Fight Against African Swine Fever


Background
The European Union is currently facing different ASF outbreak situations with front and point introductions, disease occurrence in domestic pigs and wild boar in both epidemic and endemic spread. While our traditional prevention and control strategies work usually well with industrial pig farms, we quickly reach our limits when we have to control the disease in the abundant wild boar population or in regions with a majority of backyard farms. To turn the tide and to safeguard animal health, vaccines, especially oral vaccines for wild boar, could be the missing tool. The example of classical swine fever (CSF) shows that wild boar can be successfully vaccinated against a notifiable animal disease and that control can thus be carried out quickly and efficiently. Oral vaccination of wild boar against CSF was able to reduce or even block disease introductions into the domestic pig sector. Thus, vaccines could reduce the disease burden and aid control and eradication efforts.
There has been considerable progress in vaccine development over the last years, and while we should continue to look for next generation approaches and conduct basic research, we must now also dare to test the promising candidates beyond simple proof-of-concept studies to predict their performance using established disease transmission models. Only in this way can we generate the data base for science-based benefit-risk analysis of whether and how current generation vaccines could be employed to prevent and/or control ASF.
Along these lines, the interdisciplinary ASFaVIP projet (African Swine Fever attenuated live Vaccines In Pigs) consortium sets out to a) conduct basic and applied research towards the understanding of mechanisms and correlates of protection against ASF, b) explore innovative ways to generate a next generation of ASF vaccines including generation of DIVA compatible descendants of existing vaccine viruses, c) to extensively assess promising live attenuated vaccines, in particular cell culture grown pilot vaccine batches of « ASFV-G-ΔI177L » in safety and efficacy tests after oral and intramuscular application as prescribed by international guidelines, i.e. the European Pharmacopoeia and the newly established guidelines at the World Organization for Animal Health (WOAH) and, d) verify implications to the AHL control strategy if the vaccine candidate is licensed in the EU considering possible imperfect application, uptake and protection characteristics. The overarching goal is to provide crucial data that would be required for dossier submission at the European Medicines Agency (EMA) in the process of vaccine licensing. To ensure that the regulatory affairs are dealt with in a concerted and efficient way, the consortium does not only include a strong industry partner for vaccine production (AP2, Zoetis) that has already licensed another vaccine against a category A disease with the help of a project consortium (Suvaxyn CSF Marker; CSFV_goDIVA), but also an independent scientific alliance (International Alliance for Biological Standardization, IABS-EU) that can facilitate and guide communication with stakeholders entrusted with licensure and deployment of vaccines at European and global scale. The ASFaVIP consortium will invest a major effort in characterizing the protective immune responses, also including comparison of responses against different other types of vaccines (such as efficacious vaccines, vaccines that provide only partial protection, and preparations that even result in enhanced disease course). Furthermore, the consortium will investigate host genetics influencing the disease and vaccination outcome, target genomic and serological strategies to differentiate infected from vaccinated animals (companion DIVA tests), target the optimization of oral immunization and model tailored vaccination strategies. Wild boar ecology will be considered, particularly in affected regions within the European Union. The consortium includes an associate partner from North America (AP3, USDA). This partner has developed the above-mentioned vaccine candidate and many others, accompanied by field trials in Asia, and can therefore provide the necessary critical guidance and help with innovative next generation vaccine designs, especially in the direction of DIVA concepts, and the characterization of derived strains.
Current state-of-the-art in ASF vaccines
To date, commercially available, safe and efficacious vaccines against ASF are still lacking and production of most vaccine candidates is massively hampered by the simultaneous lack of a permanent cell line that is sufficiently susceptible to ASF virus (ASFV) and which is not forcing further genetic adaptations within the ASFV genome. The STAR-IDAZ ASFV research review (2022) further mentions, amongst others, gaps regarding viral genetic patterns associated with protection, standardization and inter-laboratory testing of vaccine candidates, and different routes of vaccination. From a safety point of view, inactivated, vector or subunit vaccines could be beneficial and are therefore targeted by several research groups. They are, however, not suitable for oral vaccination of wild boar. Inactivated preparations were tested under different conditions but were not able to confer protection from disease and death. More recently, both new adjuvants and structure-preserving inactivation procedures by gamma irradiation were revisited. In both experimental approaches, high ASFV-specific antibody titers could be induced, but a protective effect was not observed. There were even indications of an acceleration of the disease process. This is consistent with the observations of other research groups, which also turned once again to the use of inactivated preparations. Based on serological responses of recovered animals, structural proteins p30 (encoded by CP204L), p54 (E183L), p72 (B646L), pp62 (CP530R) and CD2v (EP402R) have been the main targets for rational design and were tested in protein, DNA and viral vectored ASFV vaccines in challenge studies. In addition, in silico predicted antigens expressed in a variety of vector systems have been used in different projects (e.g., the ASFORCE project). Unfortunately, results with similar antigens were not always conclusive and negative results were usually not made public. The inconsistency could be attributed to a variety of factors, including the type of vaccine, vaccination strategy, the antigens, and the immune response induced, as well as the challenge model, including factors like animal genetics, virus strain, vaccine and challenge dose, and route of challenge administration. Most approaches yielded no or very limited protection. Recently, immunogenicity and efficacy of thirty-five rationally designed adenovirus vectored ASFV antigens were evaluated in wild boar. The cocktail contained adenoviruses expressing the above-named antigens and others such as EP153R, p10, p15, CP80R, I329L, H108R, K196R, CP312R, F334L, NP419L, NP868R, B66L, H339R, and R298L, which have previously been shown to be immunogenic in domestic pigs. The rest (K145R, B385R, F165R, F778R, S273R, MGF100-1L, A224L, MGF505-6R, and B175L) were selected based on the presence of putative T cell epitopes. Also here, no protection was observed upon challenge with highly virulent ASFV. However, recently, a pool of eight ASFV antigens was shown to protect pigs from lethal disease after ASFV genotype I challenge infection when vectored by a replication-deficient human adenovirus 5 (prime) and modified vaccinia Ankara (boost). The experiments were a follow-up of previous work where different antigens were shown to be immunogenic but not protective in a DNA prime/vaccinia boost approach. The protective antigen cocktail comprised gene products of B602L, B646L (p72), CP204L (p30), E183L (p54), E199L (cysteine-rich protein), EP153R (lectin-like protein), F317L, and MGF505-5R. This approach is promising in terms of antigen choice and demonstration that a DIVA compatible subunit vaccine could confer a certain level of protection. Unfortunately, and again, no clear correlates of protection could be deduced, and all animals got sick and showed considerable viraemia. Taken together, a viral-vectored vaccine against ASF is a feasible approach, however, the choice of antigens is key to success, and convincing candidates are still missing. First live vaccine approaches existed already in the 1960s. These early live vaccines were based on attenuated strains of ASFV and were used thousands of times under field conditions in both Portugal and Spain. Unfortunately, they induced chronic lesions in many vaccinated animals (even months after vaccination) and thus even led to an increase in the number of cases in both countries. Eventually, the use of the vaccine in the field was stopped. In recent years, various promising live ASF vaccine candidates have been reported that were able to induce complete or near-complete protection against challenge infection under experimental conditions. In addition to naturally occurring variants, these are mainly genetically engineered (by homologous recombination) deletion mutants that lack, in particular, genes encoding factors that bypass the host immune system. These variants have been developed and tested by several international groups, though not homogeneous in terms of dose, route and challenge. Not all of the essentially promising results have been published to date. Promising approaches with protective efficacy against currently circulating genotype II strains of ASFV include, for example, the already mentioned U.S. vaccine candidates « ASFV-G-ΔI177L » and « ASFV-G-ΔMGF », and the Chinese vaccine candidate « HLJ/18-7GD ». The naturally evolved, non-haemadsorbing virus « Lv17/WB/Rie1 » and its derivatives have also been discussed and tested as a vaccine candidate. Other viruses with deletions in different genomic regions have been described recently and are currently under further investigation. Among these recent deletion mutants with protective efficacy is the virus « ASFV-G-ΔA137R ». Other deletion mutants showed no attenuation, e.g., the virus « ASFV-G-ΔMGF110-1L ». There are now several live vaccine candidates that could be shortlisted for a potentially licensable vaccine. However, the headlines from China, which point to chronic disease courses with more respiratory and reproductive symptoms after large-scale use of illegal live vaccine candidates, urge caution in live vaccine deployment in the field prior to sufficient clinical testing. Also, based on past experience (use of classically attenuated live vaccines in Portugal and Spain), hasty solutions in ASF vaccine development are not productive. Very recently, two live vaccines have been licensed on the Vietnamese market and are now under investigation under (more or less) controlled field conditions.
Project duration
January 2024 – December 2026
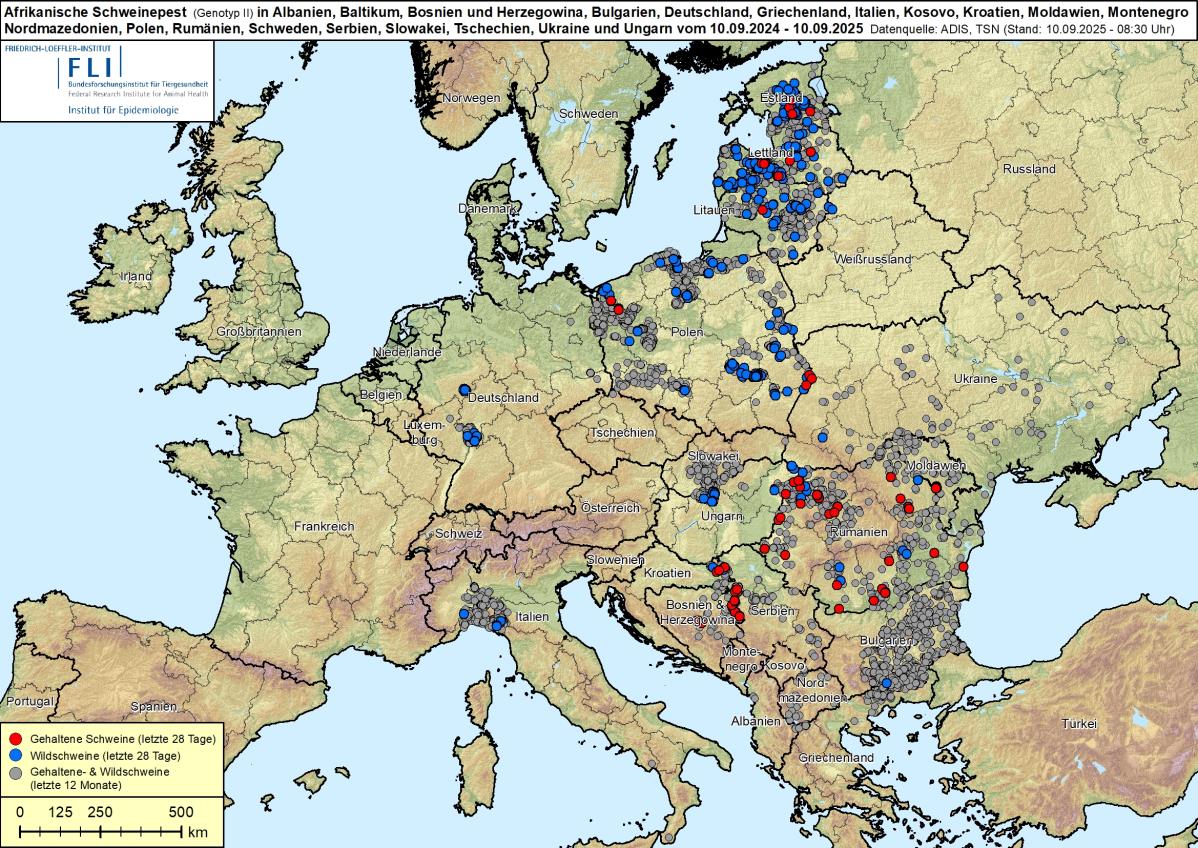
African Swine Fever in Europe: Current Outbreak Landscape